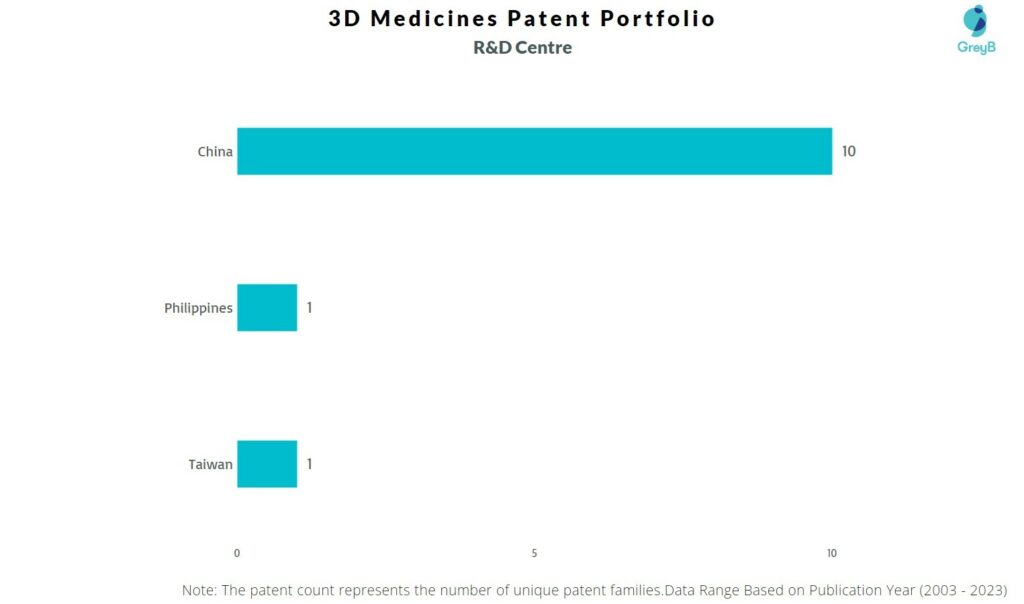
3D Medicines has a total of 43 patents globally, out of which 28 have been granted. Of these 43 patents, more than 74% patents are active. China is where 3D Medicines has filed the maximum number of patents, followed by United States of America and Japan. Parallelly, China seems to be the main focused R&D centre and also is the origin country of 3D Medicines.
3D Medicines Inc. is a biopharmaceutical company in the commercial stage that specializes in creating unique immuno-oncology drugs.
Do read about some of the most popular patents of 3D Medicines which have been covered by us in this article and also you can find 3D Medicines patents information, the worldwide patent filing activity and its patent filing trend over the years, and many other stats over 3D Medicines patent portfolio.
How many patents does 3D Medicines have?
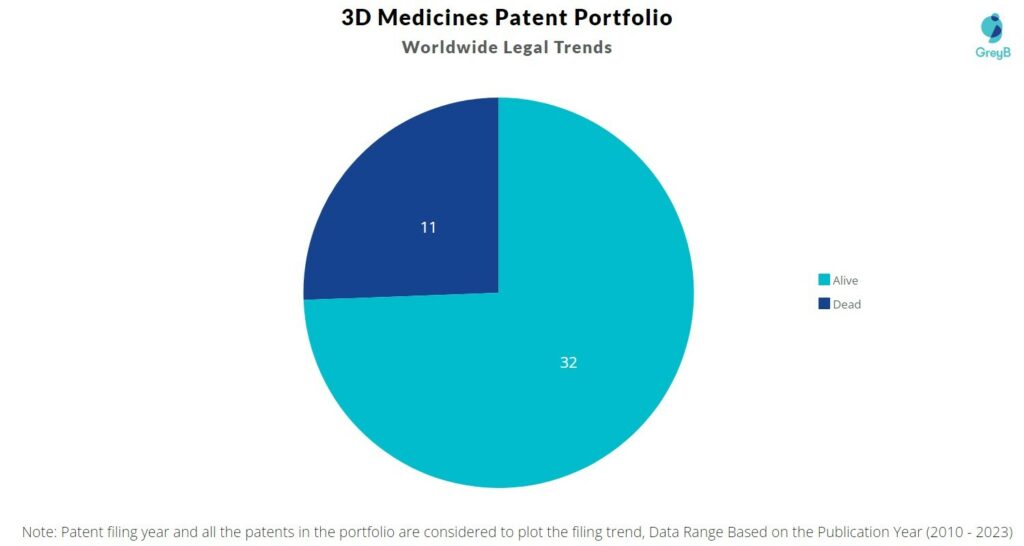
3D Medicines has a total of 43 patents globally. These patents belong to 12 unique patent families. Out of 43 patents, 32 patents are active.
How Many Patents did 3D Medicines File Every Year?
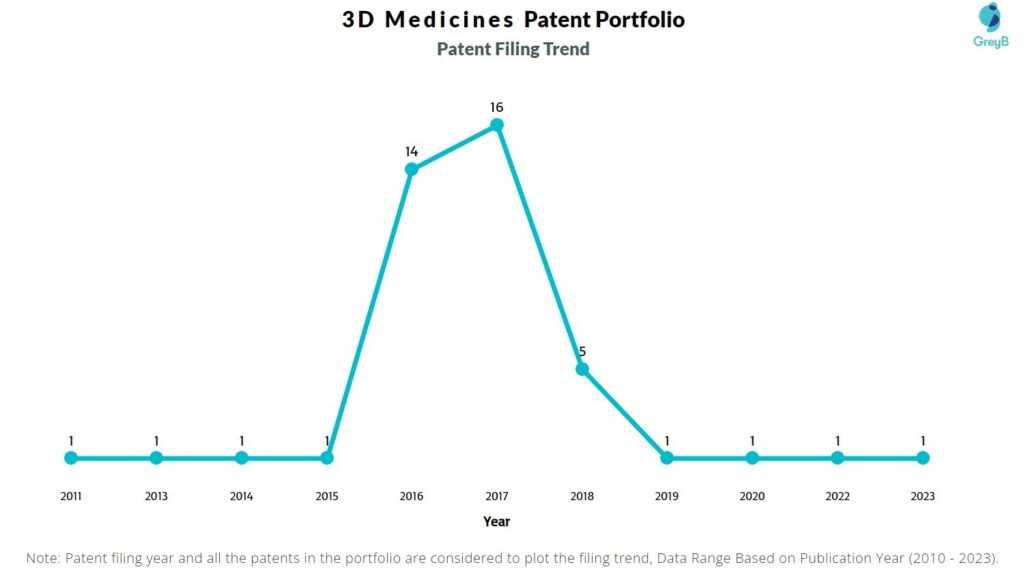
Are you wondering why there is a drop in patent filing for the last two years? It is because a patent application can take up to 18 months to get published. Certainly, it doesn’t suggest a decrease in the patent filing.
| Year of Patents Filing or Grant | 3D Medicines Applications Filed | 3D Medicines Patents Granted |
| 2023 | 1 | 6 |
| 2022 | 1 | 6 |
| 2021 | – | 4 |
| 2020 | 1 | 6 |
| 2019 | 1 | 2 |
| 2018 | 5 | 2 |
| 2017 | 16 | – |
| 2016 | 14 | 1 |
| 2015 | 1 | – |
| 2014 | 1 | 1 |
| 2013 | 1 | – |
How many 3D Medicines patents are Alive/Dead?
Worldwide Patents
How Many Patents did 3D Medicines File in Different Countries?
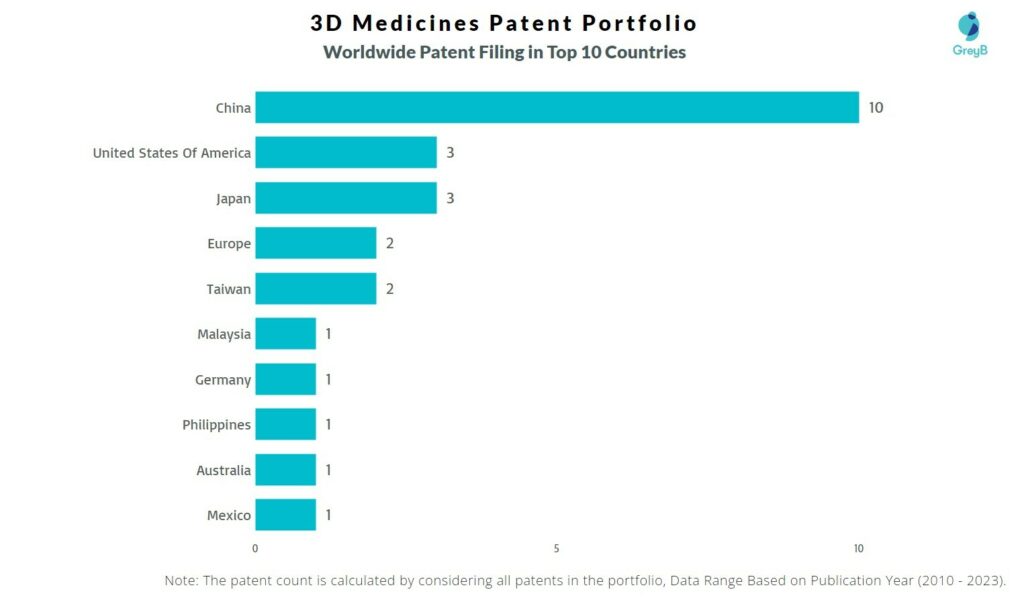
Countries in which 3D Medicines Filed Patents
| Country | Patent |
| China | 10 |
| United States Of America | 3 |
| Japan | 3 |
| Europe | 2 |
| Taiwan | 2 |
| Malaysia | 1 |
| Germany | 1 |
| Philippines | 1 |
| Australia | 1 |
| Mexico | 1 |
| Indonesia | 1 |
| New Zealand | 1 |
| Brazil | 1 |
| Russia | 1 |
| Singapore | 1 |
| Canada | 1 |
| Austria | 1 |
| Macao | 1 |
| Korea (South) | 1 |
Where are Research Centers of 3D Medicines Patents Located?
Best 3D Medicines Patents
CN107849130B is the most popular patent in the 3D Medicines portfolio. It has received 111 citations so far from company like Bj Energy Solutions.
Below is the list of most cited patents of 3D Medicines:
| Publication Number | Citation Count |
| CN107849130B | 111 |
| WO2017020802A1 | 41 |
| WO2017101789A1 | 8 |
| CN107011406A | 4 |
| WO2018090975A1 | 2 |
| WO2018090976A1 | 2 |
| KR102138447B1 | 1 |
| WO2019149219A1 | 1 |
| WO2018090974A1 | 1 |
What Percentage of 3D Medicines US Patent Applications Were Granted?
3D Medicines (Excluding its subsidiaries) has filed 3 patent applications at USPTO so far (Excluding Design and PCT applications). Out of these 1 have been granted leading to a grant rate of 50%.
Below are the key stats of 3D Medicines patent prosecution at the USPTO.
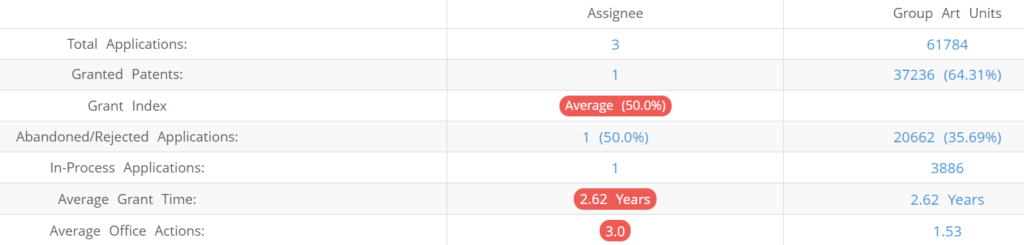
Which Law Firms Filed Most US Patents for 3D Medicines?
| Law Firm | Total Applications | Success Rate |
| Grogan Tuccillo & Vanderleeden Llp | 2 | 100% |
| Schwegman Lundberg & Woessner PA | 1 | 0% |
List of 3D Medicines patents
| 3D Medicines Patents | Title |
| US11712434B2 | Compound Having Anti-Cancer Effect, And Preparation Method Therefor And Use Thereof |
| US11351151B2 | Compound Having Anticancer Activity And Preparation Method And Application |
| EP3543228B1 | Compound Having Anticancer Activity, And Preparation Method And Application Thereof |
| EP3542796A4 | Compound Having Anti-Cancer Effect, And Preparation Method Therefor And Use Thereof |
| CN110167554B | A Compound With Anticancer Effect And Its Preparation Method And Application |
| CN110167917B | A Compound With Anticancer Effect And Its Preparation Method And Application |
| CN108350023B | A Compound With Anticancer Effect And Its Preparation Method And Application |
| CN108137644B | A Compound With Antitumor Effect And Its Preparation Method And Application |
| CN107849130B | Single Domain Antibody Against Programmed Death Ligand (Pd-L1) And Its Derivative Protein |
| CN106478760B | Triad Derivative Tba-X With Anti-Tumor Effect And Its Preparation Method And Application |
| CN105315321B | Compound With Antitumor Effect And Its Preparation Method And Application |
| CN102675401B | Preparation Of Anti-Tumor Medicine Lqc-Y And Application Thereof |
| CN116514844A | Thienopyrimidine Derivatives And Application Thereof As General Kras Mutation Inhibitor |
| WO2023138589A1 | Five-Membered Heterocyclic Pyrimidine Derivative And Use Thereof As Inhibitor Of Pan-Kras Mutation |
| JP6999960B2 | Compound Having Anti-Cancer Action, Method For Producing The Same, And Use Thereof |
| JP6993721B2 | Compound Having Anti-Cancer Activity, Method For Producing The Same, And Application Thereof |
| JP6946289B2 | Single Domain Antibody Against Programmed Death Ligand (Pdl1) And Its Derivative Protein |
| KR102138447B1 | Single Domain Antibodies For Programmed Death Ligand (Pd-L1) And Derived Proteins Thereof |
| TWI721255B | A Compound With Anticancer Effect And Its Preparation Method And Application (1) |
| TWI703134B | A Compound With Anticancer Effect And Its Preparation Method And Application (2) |
| MX402867B | Single Domain Antibody And Proteins Derived Thereof Against Programmed Death Ligand (Pdl1). |
| MY195474A | Single Domain Antibody And Derivative Proteins Thereof Against Programmed Death Ligand (Pdl1) |
| PH12018500233B1 | Single Domain Antibody Programmed Death-Ligand (Pd-L1) And Derived Protein Thereof |
| DE602017065112T2 | Anti-Cancer Compound And Method Of Manufacture And Use Thereof |
| CA2994339C | Single Domain Antibody For Programmed Death-Ligand (Pd-L1) And Derived Protein Thereof |
| IDP000074323B | Single Domain Antibody And Its Derivative Protein For Programmed Death Ligand (Pdll) |
| MOJ004079C | Single Domain Antibody Against Programmed Death Ligand (Pd-L1) And Its Derivative Protein. |
| RU2715595C2 | Single-Domain Antibody And Derivatives Thereof To Ligand-1 Protein Of Programmed Cell Death (Pdli) |
| NZ739499B | Single Domain Antibody For Programmed Death-Ligand (Pd-L1) And Derived Protein Thereof |
| AU2016302951B2 | Single Domain Antibody For Programmed Death-Ligand (Pd-L1) And Derived Protein Thereof |
| BR112018002130A2 | Single Domain Antibody And Respective Derivative Proteins Against A Programmed Death Link (Pdl1) |
| SG11201800815SA | Single Domain Antibody For Programmed Death-Ligand (Pd-L1) And Derived Protein Thereof |
| US9321804B2 | Synthesis And Use Of Anti-Tumor Drug Lqc-Y |
| CN107011406A | Anti-Tumor Compound As Well As Preparation Method And Application Thereof |
| WO2019149219A1 | Use Of Anti-Cd3 Immunotoxin Combined With Anti-Pd-L1 Single Domain Antibody In Treatment Of Cancer |
| WO2018090974A1 | Compound Having Anti-Cancer Effect, And Preparation Method Therefor And Use Thereof |
| WO2018090975A1 | Compound Having Anticancer Activity, And Preparation Method And Application Thereof |
| WO2018090979A1 | Compound Having Anticancer Effect, Preparation Method Therefor, And Applications Thereof |
| WO2018090976A1 | Compound Having Anticancer Effect, Preparation Method Therefor, And Applications Thereof |
| WO2017128981A1 | Compound Having Anti-Tumour Effect And Preparation Method And Use Thereof |
| WO2017101789A1 | Compound Having Anti-Cancer Effect, And Method For Preparation Thereof And Application Thereof |
| WO2017020802A1 | Single Domain Antibody For Programmed Death-Ligand (Pd-L1) And Derived Protein Thereof |
| AT1540404T | Verbindung Mit Antikrebswirkung Und Herstellungsverfahren Und Anwendung Davon |
What are 3D Medicines key innovation segments?
What Technologies are Covered by 3D Medicines?

The chart below distributes patents filed by 3D Medicines
