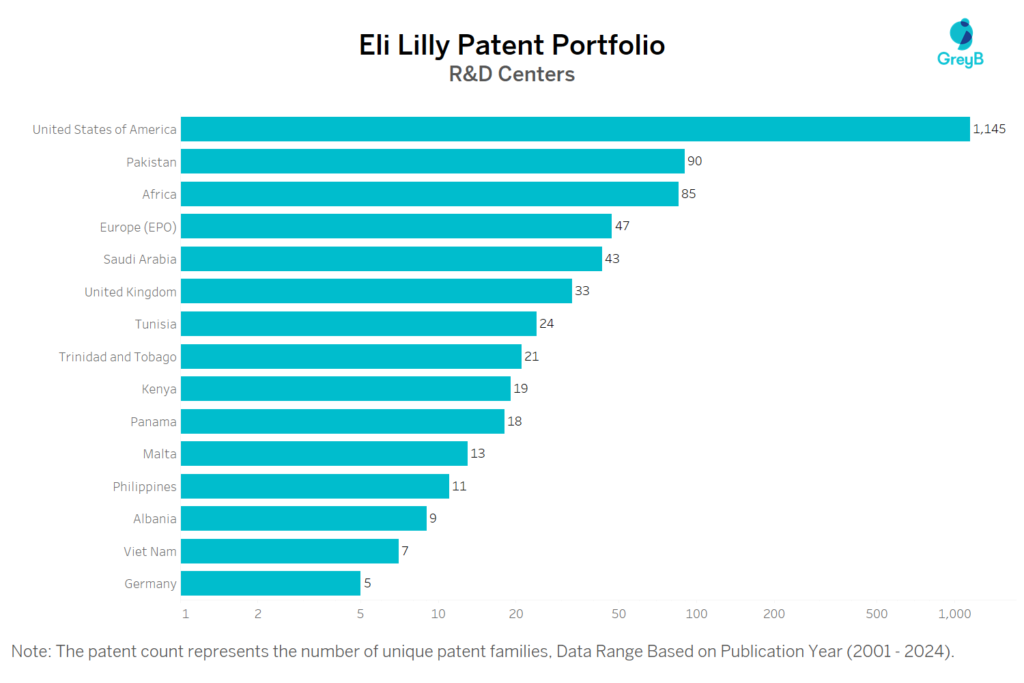
Eli Lilly has a total of 9234 patents globally, out of which 3500 have been granted. Of these 9234 patents, more than 66% patents are active. United States of America is where Eli Lilly has filed the maximum number of patents, followed by Japan and Europe. Parallelly, United States of America seems to be the main focused R&D centre and also is the origin country of Eli Lilly.
Eli Lilly was founded in the year 1876. The Company discovers, develops, manufactures, and sells pharmaceutical products for humans and animals. The Company products are sold in countries around the world. Eli Lilly products include neuroscience, endocrine, anti-infectives, cardiovascular agents, oncology, and animal health products.
Do read about some of the most popular patents of Eli Lilly which have been covered by us in this article and also you can find Eli Lilly patents information, the worldwide patent filing activity and its patent filing trend over the years, and many other stats over Eli Lilly patent portfolio.
How many patents does Eli Lilly have?
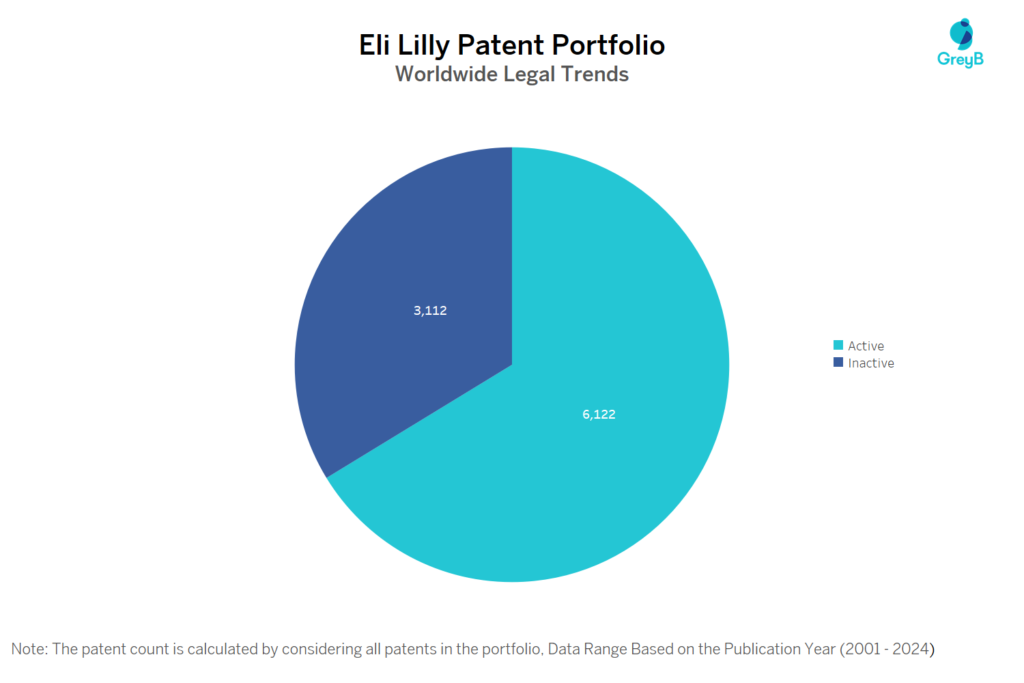
Eli Lilly has a total of 9234 patents globally. These patents belong to 1617 unique patent families. Out of 9234 patents, 6122 patents are active.
How Many Patents did Eli Lilly File Every Year?
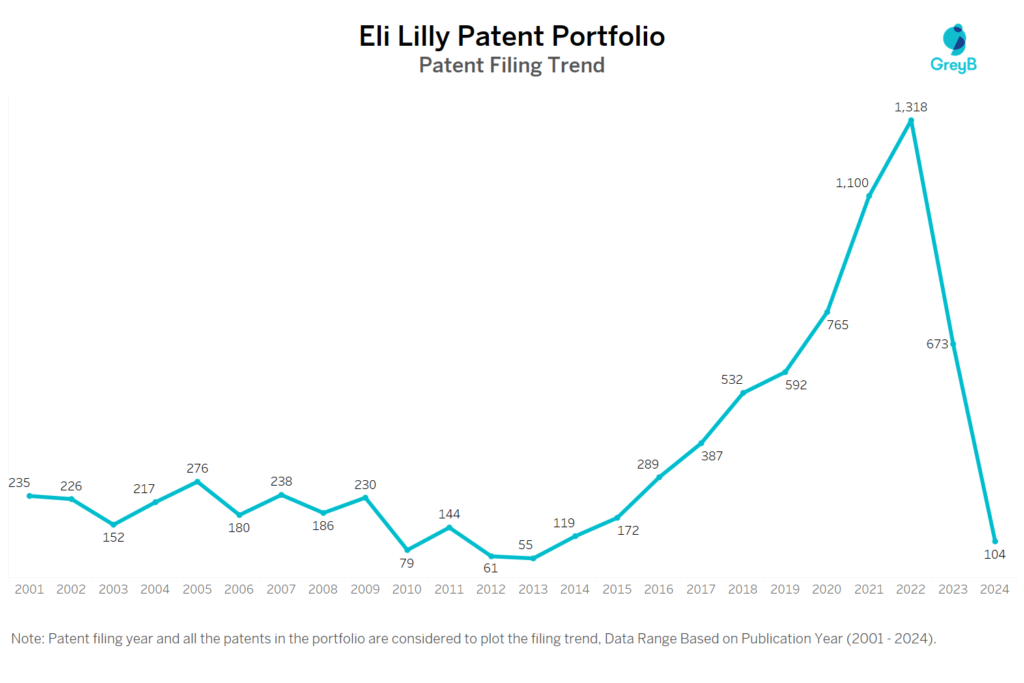
Are you wondering why there is a drop in patent filing for the last two years? It is because a patent application can take up to 18 months to get published. Certainly, it doesn’t suggest a decrease in the patent filing.
| Year of Patents Filing or Grant | Eli Lilly Applications Filed | Eli Lilly Patents Granted |
| 2024 | 104 | 440 |
| 2023 | 673 | 846 |
| 2022 | 1318 | 666 |
| 2021 | 1100 | 9 |
| 2020 | 765 | 56 |
| 2019 | 592 | 36 |
| 2018 | 532 | 32 |
| 2017 | 387 | 62 |
| 2016 | 289 | 60 |
| 2015 | 172 | 79 |
| 2014 | 119 | 95 |
| 2013 | 55 | 94 |
How many Eli Lilly patents are Alive/Dead?
How Many Patents did Eli Lilly File in Different Countries?
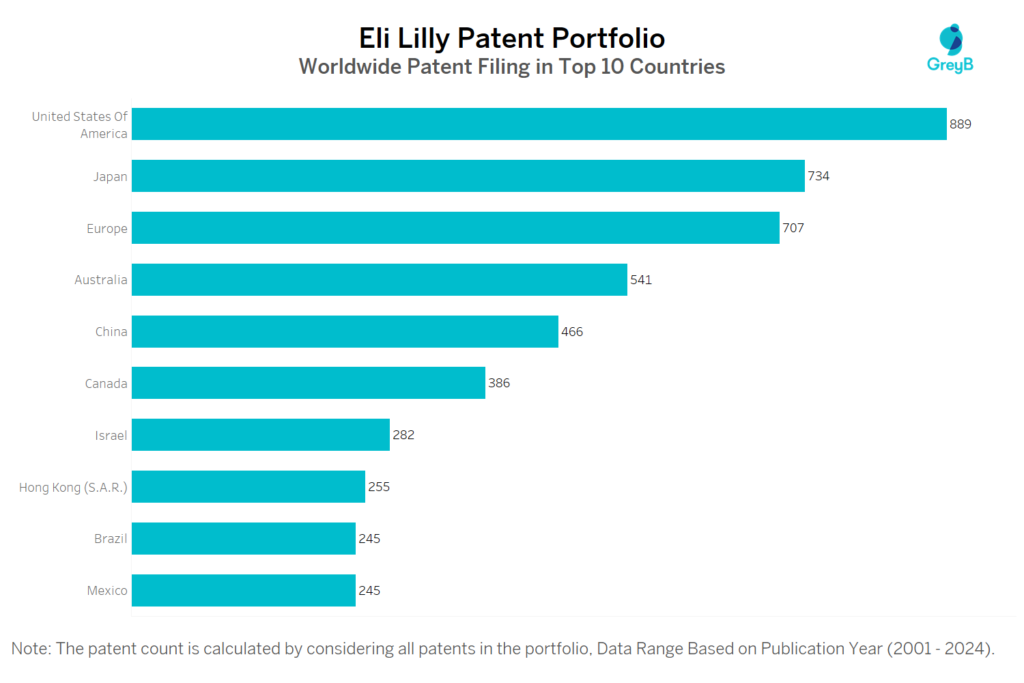
Countries in which Eli Lilly Filed Patents
| Country | Patents |
| United States Of America | 889 |
| Japan | 734 |
| Europe | 707 |
| Australia | 541 |
| China | 466 |
| Canada | 386 |
| Israel | 282 |
| Hong Kong (S.A.R.) | 255 |
| Mexico | 245 |
| Brazil | 245 |
| Germany | 227 |
| South Korea | 221 |
| India | 204 |
| Taiwan | 187 |
| Spain | 171 |
| South Africa | 171 |
| Singapore | 149 |
| New Zealand | 123 |
| Austria | 112 |
| Korea (South) | 103 |
| Argentina | 100 |
| Chile | 92 |
| Denmark | 90 |
| Pakistan | 90 |
| Africa | 86 |
| Portugal | 83 |
| Poland | 82 |
| Saudi Arabia | 78 |
| Indonesia | 75 |
| Italy | 74 |
| Peru | 70 |
| Malaysia | 67 |
| Slovenia | 64 |
| Colombia | 62 |
| Viet Nam | 62 |
| Hungary | 58 |
| Ecuador | 57 |
| Norway | 55 |
| United Kingdom | 54 |
| Cyprus | 54 |
| Costa Rica | 50 |
| Croatia | 44 |
| Philippines | 44 |
| Tunisia | 43 |
| Dominican Republic | 41 |
| Serbia | 38 |
| Ukraine | 36 |
| Lithuania | 35 |
| Macao | 34 |
| Jordan | 32 |
| Slovakia | 30 |
| Morocco | 30 |
| Russia | 24 |
| Finland | 22 |
| Trinidad And Tobago | 21 |
| Panama | 21 |
| Thailand | 20 |
| Czech Republic | 20 |
| Eurasian Patent Organization | 19 |
| Kenya | 19 |
| Malta | 13 |
| Republic Of Moldova | 13 |
| Lebanon | 10 |
| Turkey | 10 |
| Albania | 9 |
| Luxembourg | 8 |
| Uruguay | 8 |
| Estonia | 8 |
| El Salvador | 7 |
| Netherlands | 7 |
| Montenegro | 6 |
| Iceland | 5 |
| Georgia | 5 |
| Greece | 5 |
| Egypt | 4 |
| Bulgaria | 4 |
| Gulf Cooperation Council | 3 |
| Kazakhstan | 3 |
| Ireland | 2 |
| Belgium | 2 |
| United Arab Emirates | 1 |
| Botswana | 1 |
| Nicaragua | 1 |
| Romania | 1 |
| Guatemala | 1 |
| North Macedonia | 1 |
| Mongolia | 1 |
| Cuba | 1 |
| Belarus | 1 |
| Uzbekistan | 1 |
| Latvia | 1 |
| Algeria | 1 |
| Honduras | 1 |
| Brunei | 1 |
Where are Research Centres of Eli Lilly Patents Located?
10 Best Eli Lilly Patents
WO2005070963A1 is the most popular patent in the Eli Lilly portfolio. It has received 736 citations so far from companies like Xencor Inc, Macrogenics Inc and Medimmune Llc.
Below is the list of 10 most cited patents of Eli Lilly:
| Publication Number | Citation Count |
| WO2005070963A1 | 736 |
| US20040002587A1 | 587 |
| WO2005113556A1 | 568 |
| US20040102804A1 | 538 |
| WO2001027160A1 | 394 |
| US6241740B1 | 364 |
| US6800620B2 | 324 |
| US6527786B1 | 320 |
| US7288096B2 | 318 |
| US20020103345A1 | 307 |
What Percentage of Eli Lilly US Patent Applications were Granted?
Eli Lilly (Excluding its subsidiaries) has filed 3700 patent applications at USPTO so far (Excluding Design and PCT applications). Out of these 2561 have been granted leading to a grant rate of 83.86%.
Below are the key stats of Eli Lilly patent prosecution at the USPTO.

Which Law Firms Filed Most US Patents for Eli Lilly?
| Law Firm | Total Applications | Success Rate |
| Eli Lilly (Inhouse IP Department) | 2482 | 93.13% |
| Attention Patent Division | 137 | 100.00% |
| Kilpatrick Townsend & Stockton Llp | 49 | 50.00% |
| Mintz Levin Astrazeneca Ab | 40 | 72.50% |
| Elanco Us Inc | 39 | 94.87% |
| Quarles & Brady Llp Eli Lilly | 25 | 71.43% |
| Perkins Coie Llp Los General | 22 | 100.00% |
| Nielsen Ip Law Llc Eli Lilly And Company | 20 | 75.00% |
| Bristol Myers Squibb Company | 17 | 60.00% |
| David M Stemerick | 16 | 56.25% |
Lilly’s major accomplishments include becoming the first business to mass-produce Jonas Salk’s polio vaccine and insulin. Prozac, Dolophine, Cymbalta , and Zyprexa are all produced by Lilly, which is presently the largest maker of psychiatric drugs.
In 2020, the need for digital health adoption accelerated, owing in part to the COVID-19 pandemic. HCPs are urging persons with chronic illnesses to use digital services such as telehealth to continue their care when social distance and stay-at-home orders are in place, for example.
In an early-stage feasibility study, Lilly collaborated with Apple, Inc. and Evidation Health to show that an iPhone, Apple Watch, iPad, and the Beddit sleep monitoring device, in combination with digital apps, can distinguish people with mild cognitive impairment (MCI) and mild Alzheimer’s disease dementia.
Migraine pain can be so severe that in a survey recently released by Eli Lilly and Company, respondents in a survey performed by Nielsen for Eli Lilly and Company rated the worst migraine pain higher than the pain associated with kidney stones and fractured bones.
There were no migraine-specific preventive medications available until recently. Existing treatments were either difficult to tolerate or ineffective for many people. With the introduction of CGRP inhibitors, this is changing. This novel medication prevents the absorption of calcitonin gene-related peptide, or CGRP, a protein linked to migraines caused by inflammation.
EXCLUSIVE INSIGHTS COMING SOON!
What are Eli Lilly’s key innovation segments?
What Technologies are Covered by Eli Lilly?

The chart below distributes patents filed by Eli Lilly in different countries on the basis of the technology protected in patents. It also represents the markets where Eli Lilly thinks it’s important to protect particular technology inventions.

R&D Focus: How Eli Lilly search focus changed over the years?

EXCLUSIVE INSIGHTS COMING SOON!



