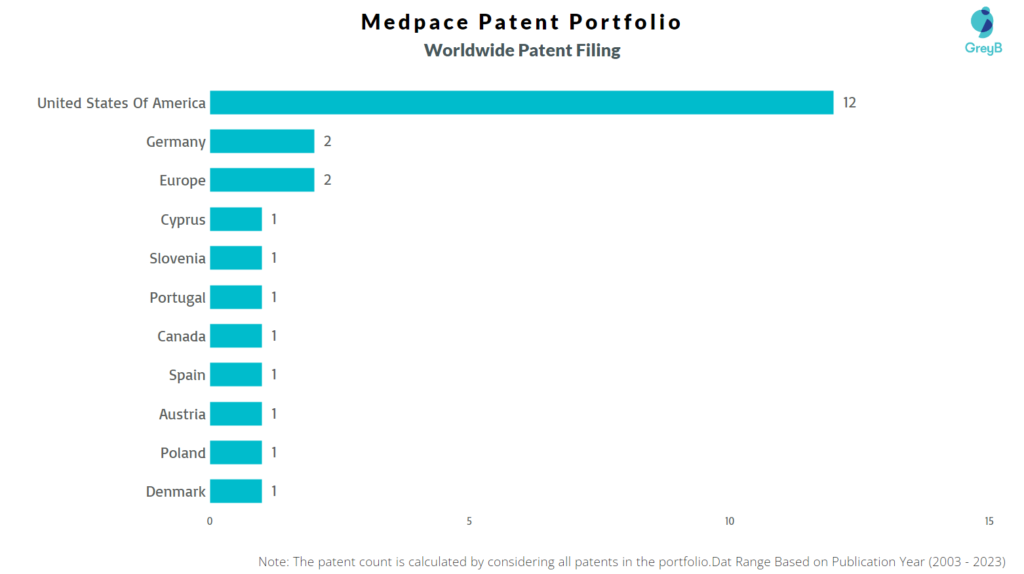
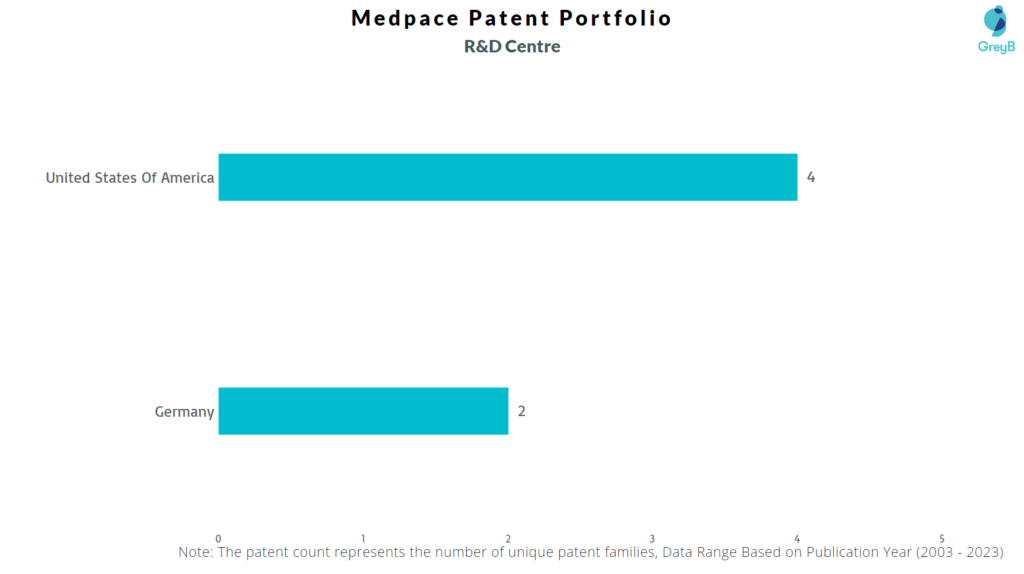
Medpace has a total of 25 patents globally, out of which 12 have been granted. Of these 25 patents, more than 4% patents are active. The USA is where Medpace has filed the maximum number of patents, followed by Europe (EPO) and Germany. Parallelly, the USA seems to be the main focused R&D center of Medpace and is also the the origin country of the firm.
Medpace was founded in the year 1992. The company is a global clinical research organization. It provides clinical development services for pharmaceutical and biotechnology industries.
Do read about some of the most popular patents of Medpace which have been covered by us in this article and also you can find Medpace patents information, the worldwide patent filing activity and its patent filing trend over the years, and many other stats over Medpace patent portfolio.
Keep track of every patent move and monitor competitor activity in real-time. Click here for full insights:
How many patents does Medpace have?
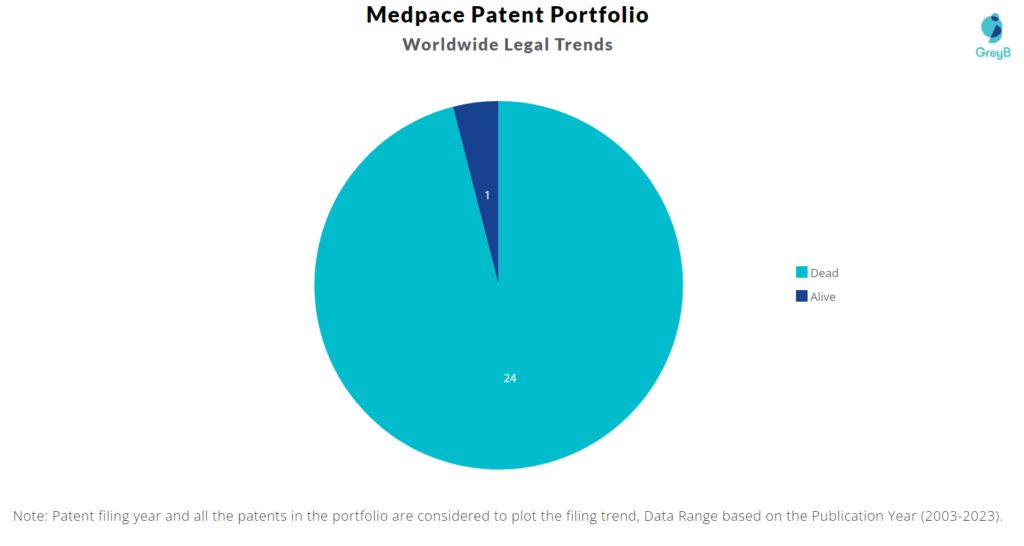
Medpace has a total of 25 patents globally. These patents belong to 6 unique patent families. Out of 25 patents, 1 patent is active.
How Many Patents did Medpace File Every Year?
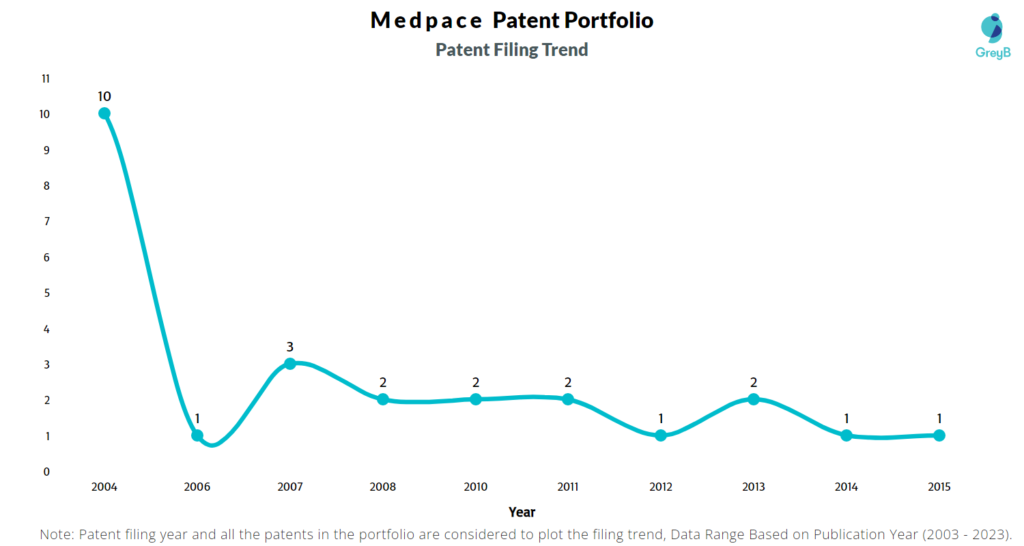
Are you wondering why there is a drop in patent filing for the last two years? It is because a patent application can take up to 18 months to get published. Certainly, it doesn’t suggest a decrease in the patent filing.
| Year of Patents Filing or Grant | Medpace Applications Filed | Medpace Patents Granted |
| 2022 | – | – |
| 2021 | – | – |
| 2020 | – | – |
| 2019 | – | – |
| 2018 | – | – |
| 2017 | – | – |
| 2016 | – | 2 |
| 2015 | 1 | 1 |
| 2014 | 1 | – |
| 2013 | 2 | 1 |
| 2012 | 1 | 6 |
| 2011 | 2 | 2 |
How many Medpace patents are Alive/Dead?
Worldwide Patents
How Many Patents did Medpace File in Different Countries?
Where are Research Centres of Medpace Patents Located?
Most Cited Medpace Patents
US20050014799A1 is the most popular patent in the Medpace portfolio. It has received 18 citations so far from companies like Cellix Bio Private Limited.
List of Medpace Patents
| Medpace Patents | Title |
| US20160045481A1 | Pyridoxamine for the Treatment of Diabetic Kidney Disease |
| US20140171472A1 | Pyridoxamine for the Treatment of Diabetic Kidney Disease |
| US20130295074A1 | Pyridoxamine for the Treatment of Diabetic Kidney Disease |
| US20130178480A1 | Inhibitors of Advanced Glycation End Products |
| US8431712B2 | Methods for the synthesis of pyridoxamine |
| US20130011379A1 | Pyridoxamine for the Treatment of Diabetic Kidney Disease |
| US8067444B2 | Pyridoxamine for the treatment of diabetic intermediaries and post-amadori inhibition |
| US20110245269A1 | Inhibitors of Advanced Glycation End Products |
| US20110178139A1 | Pyridoxamine for the Treatment of Diabetic Kidney Disease |
| US20090082407A1 | Pyridoxamine for the Treatment of Diabetic Kidney Disease |
| US20060287367A1 | Pyridoxamine for the treatment of diabetic kidney disease |
| US20050014799A1 | Pyridoxamine for the treatment of diabetic kidney disease |
| EP2041114B1 | Inhibitors of advanced glycation end products |
| EP1643999B8 | Pyridoxamine for use in the treatment of diabetic nephropathy in type ii diabetes |
| WO2008070778A2 | Pyridoxamine and low molecular weight heparinoids for diabetic kidney disease |
| CA2529538A1 | Pyridoxamine for the treatment of diabetic kidney disease |
| DE602007045052T2 | inhibitor of advanced glycation end products |
| CY1112184T1 | Pyridoxamine For Use In The Treatment Of Diabetic Nepheropathy In Type Ii Diabetes |
| PL1643999T3 | Pyridoxamine for use in the treatment of diabetic neprhopathy in type ii diabetes |
| ES2374399T3 | Pyridoxamine for the treatment of diabetic kidney disease |
| SI1643999T1 | Pyridoxamine for use in the treatment of diabetic nephropathy in type ii diabetes |
| PT1643999E | Pyridoxamine for use in the treatment of diabetic neprhopathy in type ii diabetes |
| DK1643999T3 | pyridoxamine for use in the treatment of diabetic nephropathy in type 2 diabetes |
| AT526023T | Pyridoxamin zur verwendung in der behandlung von diabetischer nephropathie bei typ ii diabetes |
| DE602004034559T2 | Pyridoxamine For Use In The Treatment Of Diabetic Nephropathy In Type Ii Diabetes |
What are Medpace key innovation segments?
What Technologies are Covered by Medpace?

The chart below distributes patents filed by Medpace
